Happy Thanksgiving to all our friends in Canada and the USA.
Enjoy the time with friends and family safely and don’t forget to visit us at NanoAndMore USA booth no. 609 @Materials_MRS MRS Fall 2021 after the holidays if you are planning to participate in the live conference.
Tag: AFM cantilevers
Molecular and nanoscale evaluation of N-cadherin expression in invasive bladder cancer cells under control conditions or GW501516 exposure
N-cadherin is a transmembrane glycoprotein expressed by mesenchymal origin cells and is located at the adherens junctions. It regulates also cell motility and contributes to cell signaling.*
A pharmacological approach to inhibit N-cadherin expression or to block its function could be relevant to prevent disease progression and metastasis development.*
In the article “Molecular and nanoscale evaluation of N-cadherin expression in invasive bladder cancer cells under control conditions or GW501516 exposure” Céline Elie-Caille, Isabelle Lascombe, Adeline Péchery, Hugues Bittard and Sylvie Fauconnet, describe how they aimed at exploring the expression level of N-cadherin in invasive bladder cancer cells upon GW501516 exposure by both molecular biology techniques such as RTqPCR and Western blotting and atomic force microscopy (AFM) using an AFM tip functionalized with a monoclonal antibody directed against this adhesion molecule. *
The Atomic Force Microscope is a mighty nanoanalytical tool for studying biological samples under liquid, in pathological or physiological conditions, and at the scale of a single cell. It allows to characterize cells and their modification upon drug exposure or function alteration, in terms of cell surface topography or cell adhesion. *
The authors demonstrated for the first time, that the PPARβ/δ activator from a concentration of 15 µM decreased the full length N-cadherin at the mRNA and protein level and significantly reduced its cell surface coverage through the measurements of the interaction forces involving this adhesion molecule. *
Using atomic force microscopy the authors carried out a morphological and topographical analysis on bladder cancer cells of different histologic grade. *
AFM imaging was carried out in contact mode on fixed cells (with an applied force of 0.1 V), the QI mode was used for alive cell imaging, all in liquid. *
Force spectroscopy in force mapping was used for cadherin/anti-cadherin antibody measurement interactions and cadherin mapping on cells. *
NanoWorld Pyrex-Nitride PNP-TR triangular shaped silicon nitride cantilevers ( CB2 with a typical spring constant of 0.08 N/m ) were used.
For force mapping the AFM cantilevers were calibrated. The AFM probes, made of silicon nitride, were functionalized by 1% APTES (3-(Aminopropyl)triethoxysilane) in toluene during 2 h, washed extensively with toluene, and then with ethanol.
The second step consisted in an incubation in 0.2% glutaraldehyde solution during 10 min, followed by extensive washing with water. A naked AFM tip was used as a negative control.
The modified AFM tips were then incubated in 50 µg/mL primary antibody solution (N-cadherin GC-4 clone directed against the extracellular domain, N-cadherin 3B9 clone directed against the intracellular domain, E-cadherin HECD-1 clone directed against the extracellular domain) during 30 min, then washed with PBS 1X.
Finally, the functionalized AFM tip was saturated by incubation in 2 mg/mL RSA (rat serum albumin) solution during 30 min. *
Quantitative imaging AFM mode enabled to register more than hundred force spectroscopy curves per condition. The curves registered on cells were overlayed in order to highlight a specific pattern and the interaction peak areas were measured. *

T24 and RT4 bladder cancer cell morphology and topography. a Images from control confluent cells by phase contrast microscopy. Scale bars: 200 µm. b, c AFM images obtained on control confluent cells, after glutaraldehyde fixation, in contact mode in liquid. b AFM height images. c AFM deflection images. Scale bars: 10 µm
* Céline Elie-Caille, Isabelle Lascombe, Adeline Péchery, Hugues Bittard amd Sylvie Fauconnet
Molecular and nanoscale evaluation of N-cadherin expression in invasive bladder cancer cells under control conditions or GW501516 exposure
Molecular and Cellular Biochemistry (2020) 471:113–127
DOI: https://doi.org/10.1007/s11010-020-03771-1
Please follow this external link to read the full article: https://link.springer.com/article/10.1007/s11010-020-03771-1
Open Access : The article “Molecular and nanoscale evaluation of N-cadherin expression in invasive bladder cancer cells under control conditions or GW501516 exposure” by Céline Elie-Caille, Isabelle Lascombe, Adeline Péchery, Hugues Bittard and Sylvie Fauconnet is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit https://creativecommons.org/licenses/by/4.0/.
Human ESCRT-III polymers assemble on positively curved membranes and induce helical membrane tube formation
The Endosomal Sorting Complex Required for Transport-III (ESCRT-III) is part of a conserved membrane remodeling machine. ESCRT-III employs polymer formation to catalyze inside-out membrane fission processes in a large variety of cellular processes, including budding of endosomal vesicles and enveloped viruses, cytokinesis, nuclear envelope reformation, plasma membrane repair, exosome formation, neuron pruning, dendritic spine maintenance, and preperoxisomal vesicle biogenesis.*
How membrane shape influences ESCRT-III polymerization and how ESCRT-III shapes membranes is yet unclear.*
In the article “Human ESCRT-III polymers assemble on positively curved membranes and induce helical membrane tube formation” Aurélie Bertin, Nicola de Franceschi, Eugenio de la Mora, Sourav Maity, Maryam Alqabandi, Nolwen Miguet, Aurélie di Cicco, Wouter H. Roos, Stéphanie Mangenot, Winfried Weissenhorn and Patricia Bassereau describe how human core ESCRT-III proteins, CHMP4B, CHMP2A, CHMP2B and CHMP3 are used to address this issue in vitro by combining membrane nanotube pulling experiments, cryo-electron tomography and Atomic Force Microscopy.*
The authors show that CHMP4B filaments preferentially bind to flat membranes or to tubes with positive mean curvature.*
The results presented in the article cited above underline the versatile membrane remodeling activity of ESCRT-III that may be a general feature required for cellular membrane remodeling processes.*
The authors provide novel insight on how mechanics and geometry of the membrane and of ESCRT-III assemblies can generate forces to shape a membrane neck.*
NanoWorld Ultra-Short AFM Cantilevers USC-F1.2-k0.15 were used for the High-speed Atomic Force Microscopy ( HS-AFM ) experiments presented in this article.*
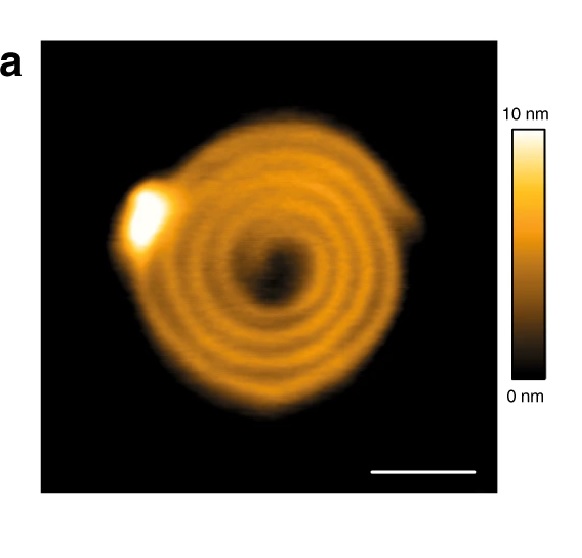
CHMP4-ΔC flattens LUVs and binds preferentially to flat membranes or to membranes with a positive mean curvature.
1a CHMP4B-ΔC spirals observed by HS-AFM on a lipid bilayer. Scale bar: 50 nm.
Please refer to the full article for the complete figure: https://rdcu.be/b5rOe
*Aurélie Bertin, Nicola de Franceschi, Eugenio de la Mora, Sourav Maity, Maryam Alqabandi, Nolwen Miguet, Aurélie di Cicco, Wouter H. Roos, Stéphanie Mangenot, Winfried Weissenhorn and Patricia Bassereau
Human ESCRT-III polymers assemble on positively curved membranes and induce helical membrane tube formation
Nature Communications volume 11, Article number: 2663 (2020)
DOI: https://doi.org/10.1038/s41467-020-16368-5
Please follow this external link to read the full article: https://rdcu.be/b5rOe
Open Access The article “ Human ESCRT-III polymers assemble on positively curved membranes and induce helical membrane tube formation “ by Aurélie Bertin, Nicola de Franceschi, Eugenio de la Mora, Sourav Maity, Maryam Alqabandi, Nolwen Miguet, Aurélie di Cicco, Wouter H. Roos, Stéphanie Mangenot, Winfried Weissenhorn and Patricia Bassereau is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.

